Clinical trials are now a global activity, as you can appreciate by one slide presented during a recent WHO webinar and reported here. The WHO developed a guideline focusing on the most critical issues which can be faced in implementing new clinical trials in various regions of the world.
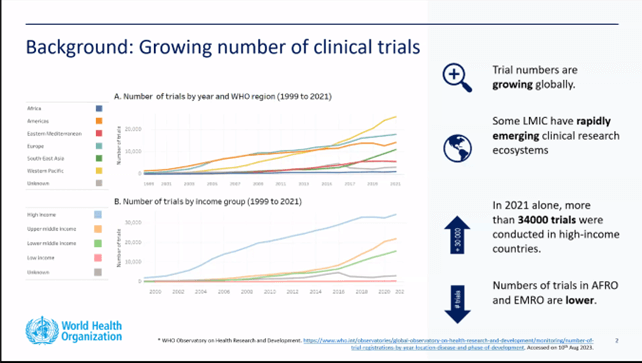
The new guideline was presented during a webinar held on September 25 and attended by more than 3000 professionals from all over the world: there were several contributions from various authors who underlined the importance of gender equity, attention to rare diseases, but above all, the importance to plan for well designed clinical studies, in order to obtain meaningful results.1
You can read the new guideline here.
References
- Launch of guidance for best practices for clinical trials. (2024, September 25). https://www.who.int/news-room/events/detail/2024/09/25/default-calendar/launch-of-guidance-for-best-practices-for-clinical-trials
Disclaimers
- The material in these reviews is from various public open-access sources, meant for educational and informational purposes only
- Any personal opinions expressed are those of only the author(s) and are not intended to represent the position of any organization(s)
- No official support by any organization(s) has been provided or should be inferred


















