It was only a few years ago that patients and healthcare professionals were concerned that the FDA approvals of new drugs reached a nadir of fewer than 20 NCEs (new chemical entities). Thankfully, this situation saw a dramatic change in the last decade, and we in the pharmaceutical world are happy to point out that, in 2021, the FDA approved a record number of 50 NCEs! Clearly, scientists are gaining a better understanding of several diseases and this is the solid foundation on which NCEs with effective mechanisms of action are designed.
Below you will find graphs featuring the therapeutic areas of the 50 new approvals, and some of their common characteristics.
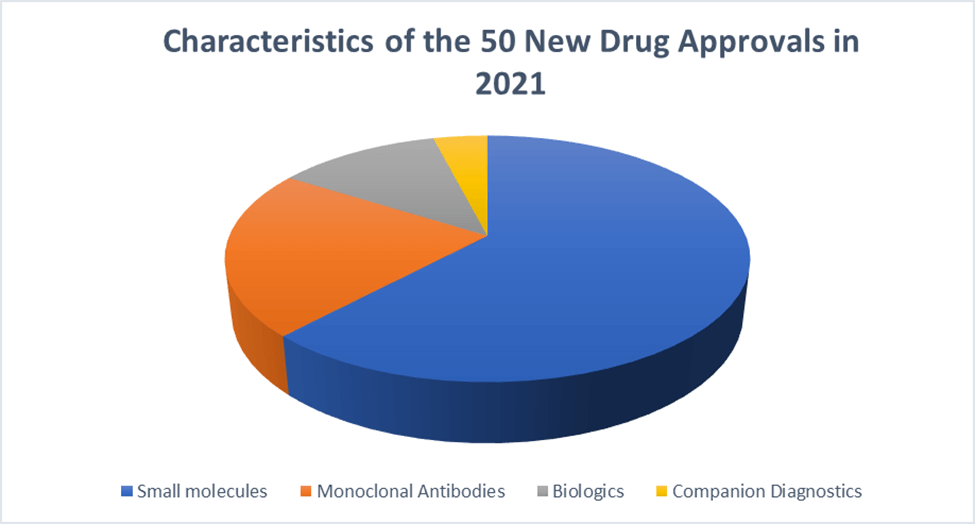
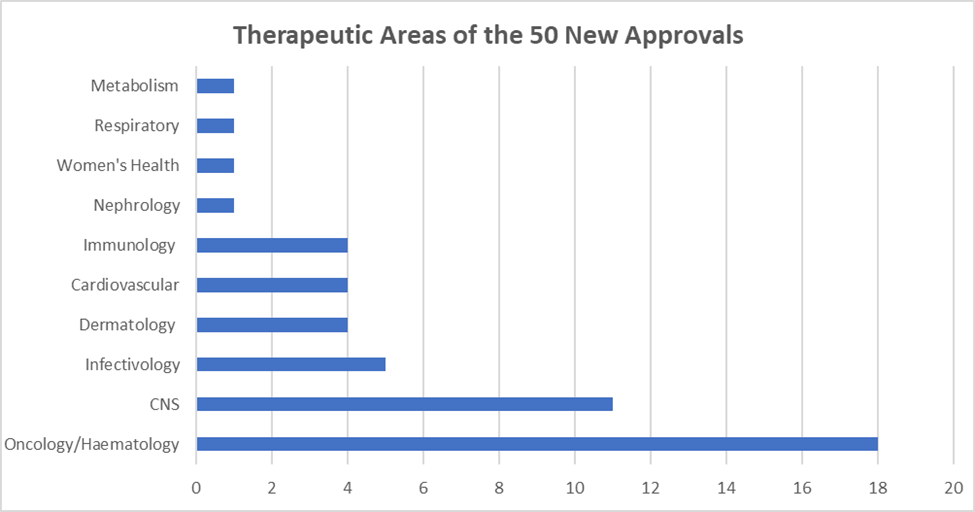
Some takeaways from the data:
- Small molecules represent about two-thirds of the approvals.
- The era of biologics seems to be replaced by the era of monoclonal antibodies.
- Note the presence of two companion diagnostics.
- Oncology and hematology are the two top therapeutic areas.
- CNS is also largely represented.
- The cardiovascular system, despite the fact that it holds the first position of events causing morbidity and mortality, has only 4 new entries.
Disclaimers
- The material in these reviews is from various public open access sources, meant for educational and informational purposes only
- Any personal opinions expressed are those of only the author(s) and are not intended to represent the position of any organization(s)
- No official support by any organization(s) has been provided or should be inferred


















