Pharmaceutical quality is a must! We all know that, in the pharmaceutical world, we have three fundamental pillars: quality (outlined in the GMP guidelines), safety (which refers to GLP), and efficacy (based on GCP).
With the opportunities offered by globalization, the responsibilities of both pharmaceutical companies and regulatory agencies have significantly increased, and audits and inspections of manufacturing sites represent a significant investment in time and human resources.
Every year the FDA prepares a report about pharmaceutical quality, outlining major changes in the manufacturing scenario, and key findings from their inspections performed both in the USA and abroad.
Figure 1 of the report includes a snapshot of changes made to the world of drug manufacturing.
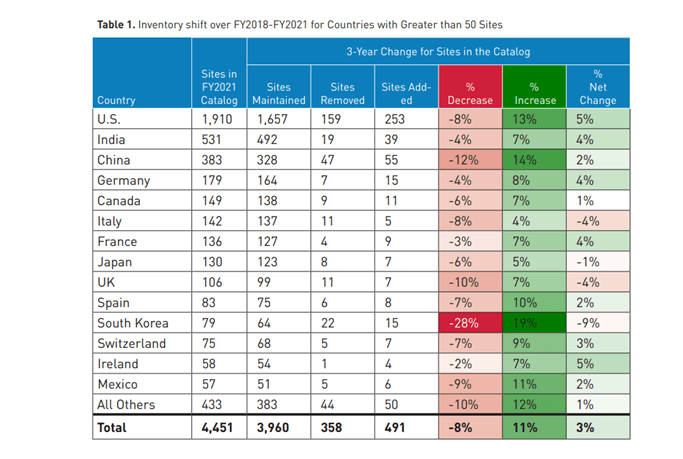
You can read the full report here.
Disclaimers
- The material in these reviews is from various public open access sources, meant for educational and informational purposes only
- Any personal opinions expressed are those of only the author(s) and are not intended to represent the position of any organization(s)
- No official support by any organization(s) has been provided or should be inferred


















