In an effort to educate both healthcare professionals and patients to expand the use of biosimilars, the FDA has posted several pages dedicated to this family of medicinal products.
Biosimilars approved by the FDA are safe and effective biological medications. The availability of biosimilars can provide patients with more treatment options, increase access to lifesaving medications, and potentially lower healthcare costs through competition.
Biological products are used to treat many illnesses, including chronic skin diseases, such as psoriasis; inflammatory bowel diseases, such as Crohn’s disease and ulcerative colitis; arthritis; kidney conditions; diabetes; and cancer. Biologics are generally large, complex molecules that are made from living sources such as bacteria, yeast, and animal cells. Because they generally come from living organisms, biologics inherently contain many slight variations from batch to batch, and their structures are generally more complex than those of other medications. As a result, biologics are often more complicated to purify, process, and manufacture. There are many types of biologics approved for use in the United States, such as monoclonal antibodies, insulin, vaccines, and allergenic products.1
Read more here.
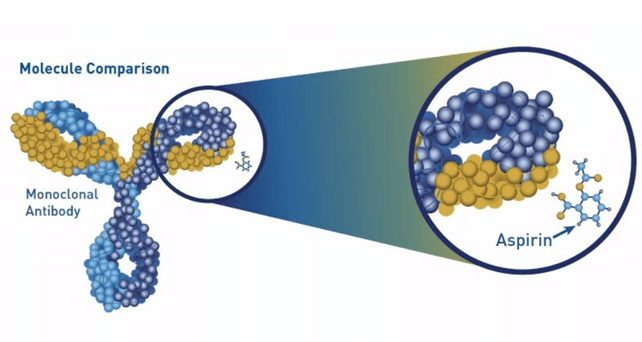
The image below illustrates the significant difference in size between aspirin and a monoclonal antibody.
References
- Research Overview for Health Care Professionals. U.S. Food And Drug Administration. https://www.fda.gov/drugs/biosimilars/overview-health-care-professionals
Disclaimers
- The material in these reviews is from various public open-access sources, meant for educational and informational purposes only
- Any personal opinions expressed are those of only the author(s) and are not intended to represent the position of any organization(s)
- No official support by any organization(s) has been provided or should be inferred


















