We recently reported changes in guidelines regarding the use of aspirin for the prevention of cardiovascular disease. Any change in well-established guidelines may offer the opportunity for experts to engage in a scientific debate to raise and address key components, as well as improve the understanding of the new guideline.
The Academy asked Professor Soneil Guptha, an experienced cardiologist and a senior member of the Academy, to personally comment on the new guideline. We are pleased to offer his opinion and welcome additional comments from all of you.
Aspirin for CV Risk Reduction
Soneil Guptha MD FACC FESC FCCP FICA MFPM GFMD
Domenico Criscuolo MD PhD GFMD
DISCLAIMER
The commentary by the authors are opinions synthesized from the understanding of the publicly available science and research material and does not represent the position of any organization they have worked for.
The content is meant for a thoughtful critique rather than any guideline to be followed.
Commentary:
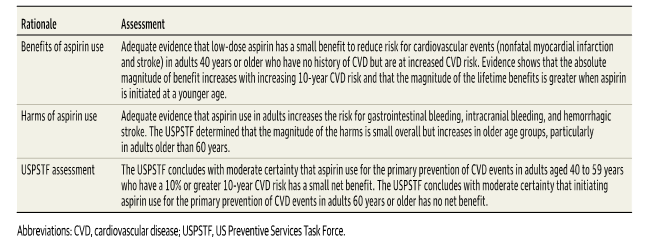
Recent United States Preventive Services Task Force (USPSTF) recommendations [JAMA.2022;327(16):1577-1584. doi:10.1001/jama.2022.4983] for cardiovascular disease (CVD) prevention can be summarized as follows. Summary of recommendations:
Source: [JAMA.2022;327(16):1577-1584. doi:10.1001/jama.2022.4983]

Summary of USPSTF Rationale Source: [JAMA.2022;327(16):1577-1584. doi:10.1001/jama.2022.4983]
The holy grail of preventive medicine is that benefit must outweigh the harm. If one subscribes to that commandment, then it makes sense not to administer a medicine when the subject is healthy or is at high risk of harm for any reason (inclusive of their inherent genetics or concurrent ailments). Given that the second statement stands true to the statuary Hippocratic Oath or the fiduciary responsibility of the health care practitioner.
The conundrum is to tease the evidence and make an informed decision on who would benefit. The best way in the development of evidence is by a randomized control trial. In today’s world it would be not possible to conduct a population-based randomized controlled placebo control trial with aspirin (only) for many reasons viz. large sample size, high cost, diverse global management/treatment practices or guidelines, high variability of background/epidemiological natural history of the diseases, confounding illnesses, and lifestyle choices of non-pharmacological management of health, amongst the long list of variables that could introduce the biases in such a trial and make the understanding of results complex/complicated.
Given this, the experts have two choices. (1) one is to synthesize the evidence with statistical modeling/metanalysis/systematic reviews/pooled analyses from existing results; despite the complexities and controversies of these methodologies or (2) conduct a multicenter global trial with a modest sample population whilst addressing the pharmacological and non-pharmacological preventative treatment strategies and piggyback a research question with a placebo control trial (preferably double-blinded). The USPSTF systematic review on aspirin effectiveness in the reduction of CVD events (myocardial infarctions and strokes), CV mortality, and all-cause mortality in subjects with no history of CVD utilizes the first approach. This systematic analysis also included a microsimulation modeling study (for primary prevention only) to assess the net benefit and harm from aspirin; stratified for age, gender, and CVD risk level.
Lessons:
The evidence from both approaches is self-explanatory. As pharmaceutical physicians, developers of medicines and medical affairs executives, the responsibility of generating evidence has been very high for regulatory approvals. However, in current and future times, the focus for precision medicine and targeted approach for informed real-world evidence generation and evidence-informed practice (which considers the patient’s agreement in treatment/management) along with a deeper understanding of statistical methods and modeling is to be/will be expected. Thus, Pharmaceutical Physicians/Drug Development Scientists/Medical Affairs professionals must ensure understanding of statistical methodology; risk-benefit ratios; health economic paradigms, the advantages, and limitations of conducting systematic reviews or metanalysis and the generalizability or specific applicability of inferences drawn. The following teases the above and elaborates a bit more. (It must be understood that details clinical design and statistical methodology discussion is beyond the scope of this review).
- The USPSTF systematic review is US/region-centric.
- The epidemiology of heart disease morbidity and mortality has regional/population variability. Thus, evidence generation and implementation must consider the ethnic and population/genetic and migratory secular trends of the disease and disease management practices.
- The epidemiology of heart disease morbidity and mortality has regional/population variability. Thus, evidence generation and implementation must consider the ethnic and population/genetic and migratory secular trends of the disease and disease management practices.
- The USPSTF systematic review addresses the balance of benefit and harm and could benefit with some estimations and projections of the costs (effectiveness, utility vis-à-vis benefit and harm).
- More research is needed to evaluate the following. • Improving the accuracy of CVD risk prediction in all racial and ethnic and socioeconomic groups. • The gastrointestinal bleeding risk associated with aspirin use in populations representative of the US primary CVD prevention population. • Characterizing the distribution of patient preferences across the spectrum of cardiovascular risk after patients are informed about the benefits and harms of aspirin. • The effects of low-dose aspirin use on CRC incidence and mortality over the long term (10 to 20 years and longer).
Concluding Remarks:
- Common sense and evidence-based on collective experiences, research designs, statistics, statistical modeling, and health economics must become more entrenched and enshrined to ensure that the results have high internal and external (dual) validity.
- Genetics, patient-centered research (study designs) and real-world evidence generation is expected to change paradigms for medicines development success in the next decade or beyond.
Recent Publications of Interest
- Wang M, Yu H, Li Z, Gong D, Liu X. Benefits and Risks Associated with Low-Dose Aspirin Use for the Primary Prevention of Cardiovascular Disease: A Systematic Review and Meta-Analysis of Randomized Control Trials and Trial Sequential Analysis. Am J Cardiovasc Drugs. 2022 May 16. doi: 10.1007/s40256-022-00537-6.
- Li J, Chen Y, Ou Z, Ouyang F, Liang J, Jiang Z, Chen C, Li P, Chen J, Wei J, Zeng J. Aspirin Therapy in Cardiovascular Disease with Glucose-6-Phosphate Dehydrogenase Deficiency, Safe or Not? Am J Cardiovasc Drugs. 2021 Jul;21(4):377-382. doi: 10.1007/s40256-020-00460-8.
- Fras Z, Sahebkar A, Banach M. The Use of Aspirin in Contemporary Primary Prevention of Atherosclerotic Cardiovascular Diseases Revisited: The Increasing Need and Call for a Personalized Therapeutic Approach. Am J Cardiovasc Drugs. 2021 Mar;21(2):139-151. doi: 10.1007/s40256-020-00424-y.
- Mainoli B, Duarte GS, Costa J, Ferreira J, Caldeira D. Once- versus Twice-Daily Aspirin in Patients at High Risk of Thrombotic Events: Systematic Review and Meta-Analysis. Am J Cardiovasc Drugs. 2021 Jan;21(1):63-71. doi: 10.1007/s40256-020-00409-x


















